RT630-HT01 Drug Dissolution Testing Equipment
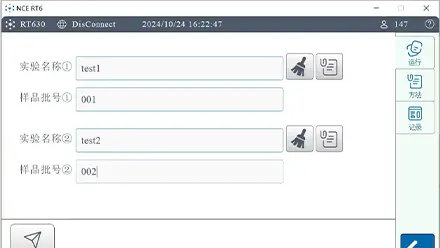
The RT630-HT02 Drug Dissolution Testing Equipment operates as a precise dual-module automatic sampling system which satisfies all requirements from Chp (0931), USP (711/724), and EP (2.9.3/4). The device runs two autonomous dissolution tests simultaneously which supports distinct devices and speeds and solvents and sampling points for optimized testing results
Learn More
Get Free Quote
Download Sheet
- Dual-mode Dissolution System
- All-in-one Dual-mode Sampler
- Built-in Thermostat Rehydration Cups
- Innovative Design
-
Description
-
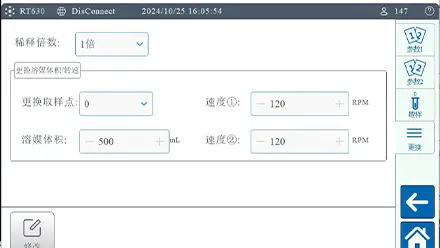
The device contains real-time monitoring technology together with independent temperature probes which precisely monitor the solvent temperatures in 12 separate vessels. The high-precision sampling pump teamed with automatic online filtering minimizes sampling inaccuracies and stops residual contamination from happening. Adjustments of both dissolution medium volume and rotational speed through the device’s advanced design meet method requirements.
The RT630-HT02 Drug Dissolution Testing Equipment operates as a precise dual-module automatic sampling system which satisfies all requirements from Chp (0931), USP (711/724), and EP (2.9.3/4). The device runs two autonomous dissolution tests simultaneously which supports distinct devices and speeds and solvents and sampling points for optimized testing results.
Features
Application
Basket and paddle methods are the most commonly used legal dissolution methods in national pharmacopoeias and were the first dissolution methods introduced into pharmacopoeias. They are mainly used for quality control (QC) of pharmaceutical products.
-
Oral Tablets
-
Patch
-
Semi-solid preparations
-
Injection
-
Intrinsic Dissolution Rate of API
Specifications
Product Brochure

General Dissolution System
|
Length ×Width× Height (Fully Raised): 95cm × 65cm ×82 cm |
Dissolution Vessel Number: 14 (12 dissolution levels+2 thermostatic refill levels) |
|
Setting Range: 20-250 rpm (RPM) |
RPM Resolution: 0.01RPM |
|
Steady Speed Error: ≤±0.3RPM |
Temp. Accuracy: ≤±0.2℃ |
|
Temp. Resolution: 0.01 ℃ |
Level: < 0.5° |
|
Vessel Verticality: 90°±0.5° |
Shaft Verticality: 90°±0.5° |
|
Centering Deviation: < ±2.0 mm |
Depth Positioning Deviation: < ±1.0 mm |
|
Shaft Wobble: < ±1.0 mm |
Basket Wobble: < ±1.0 mm |
Automatic Sampling Workstation
|
Length ×Width× Height: 40 cm × 70cm ×50 cm |
Maximum Settable Sample Number: 36 |
|
Sampling Range: 1 ~ 20ml |
Minimum Sampling Interval: 4 minutes |
|
Maximum Working Time: 720 hours |
Sampling Precision: ≤±1% |
Operating System
|
Meeting FDA21 CFR Part 11 |
Have A Business Inquiry?
Interested to find out more about how our products and services can serve your business needs? Drop us an online inquiry and our qualified professionals will reach out to you.
Get Free Quote