Suspending Eye Drops

Ophthalmic suspension is medical eye drops that are characterized by the presence of incompletely dissolved solid particles. These particles are evenly dispersed in a liquid matrix, allowing the medication to provide a sustained release when dropped into the eye.
Get Free Quote
Experimental parameters
| Dissolution apparatus | RT7 Flow-through Cell Dissolution System |
| Temperature | 37℃± 0.5℃ |
| Model | Closed-loop |
Experimental Result
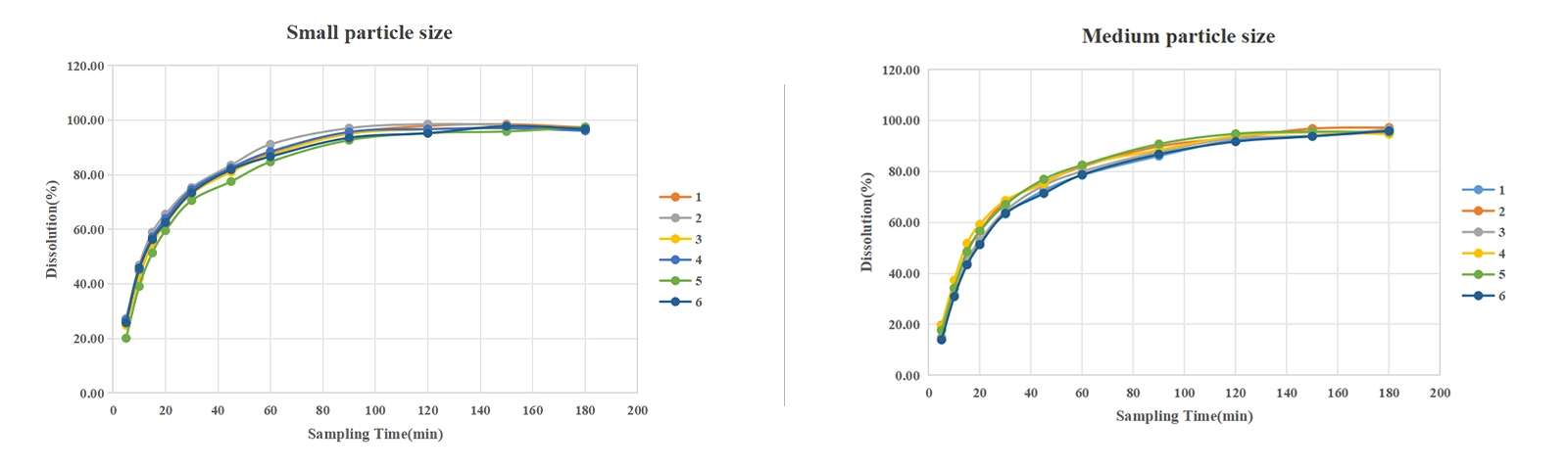
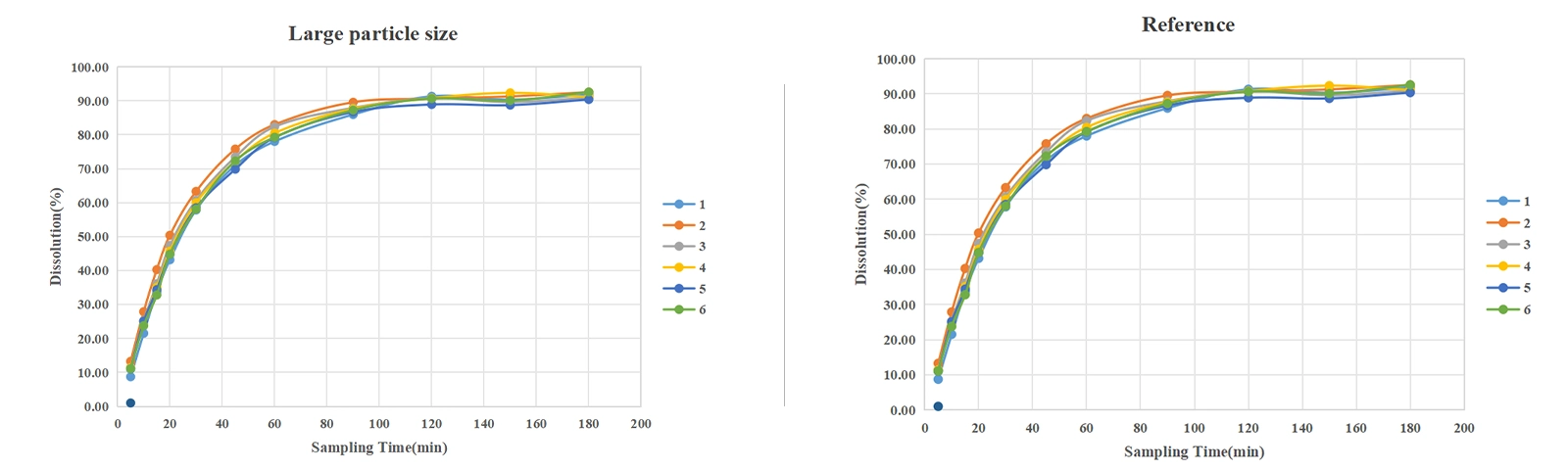
①Reproducibility: The RSD ranges were 0.51~10.44%, 0.95~13.58%, 0.91~12.99%, and 0.72~14.93% for small, medium, large particle size, and reference, respectively.
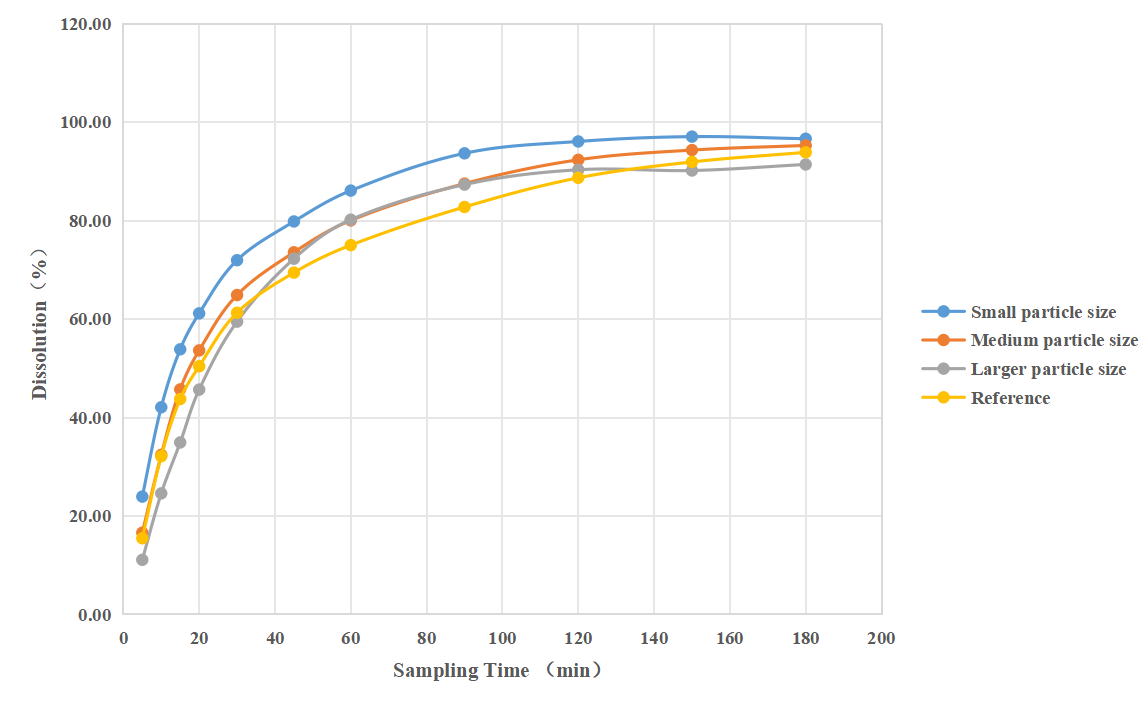
②Distinguish: This method was able to demonstrate the effect of changes in CQA (particle size) on the in vitro release behavior of this ophthalmic suspension. And assist in differentiating between generic and reference formulations.
CONCLUSION: RT700 provides good reproducibility and distinguish for in vitro dissolution testing of ophthalmic suspension

